ANCA-associated vasculitis causes
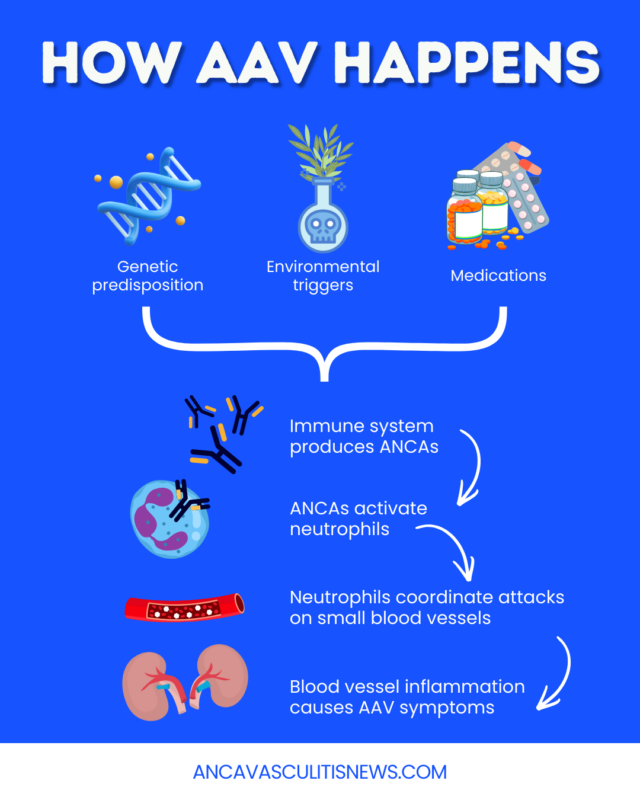
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) encompasses a group of diseases characterized by inflammation and damage in the body’s small blood vessels. Although scientists don’t know precisely what causes AAV, genetic and environmental factors likely contribute.
These interacting elements, in ways that are incompletely understood, may trigger the production of ANCAs — a group of self-reactive antibodies that usually activate the abnormal immune response seen in AAV. The resulting small blood vessel inflammation causes a range of symptoms depending on the affected organs or tissues.
The role of ANCAs
ANCAs are thought to directly drive abnormal immune reactions for most people with AAV. These antibodies bind to proteins on immune cells called neutrophils. This activates the neutrophils, which then release harmful substances that damage healthy blood vessel cells.
Abnormal neutrophil signaling also recruits other immune cells to the area, promoting further ANCA production and exacerbating the inflammatory response.
ANCAs generally target one of two neutrophil proteins:
- myeloperoxidase (MPO)
- proteinase 3 (PR3)
The specific ANCA target is closely associated with the type of AAV. MPO-ANCAs are usually related to microscopic polyangiitis (MPA), while PR3-ANCAs are more often associated with granulomatosis with polyangiitis (GPA).
Some individuals, including many with the rarest type of AAV, called eosinophilic granulomatosis with polyangiitis (EGPA), have neither typical ANCA type. Some may have antibodies that target other neutrophil proteins, although this remains incompletely understood.
Genetic factors
AAV is not inherited, meaning it isn’t caused by specific genetic mutations that are passed down in families. However, research suggests that genetic variants related to immune function could influence a person’s risk of developing AAV and the ANCA type they have.
Still, genetic factors likely interact with various environmental factors to influence disease risk in ways that are not fully understood. As such, doctors can’t use genetics or family history alone to determine:
- the specific probability of developing AAV
- symptoms or prognosis
- AAV treatment responses
Likewise, because AAV is not solely a genetic disorder, a lack of family history of vasculitis does not rule out the disease.
Environmental and external triggers
Other environmental factors may also increase the long-term risk of AAV, including chronic exposure to:
- silica dust, found in sand, soil, rock, and human-made materials like concrete and plaster
- air pollution
- organic solvents and certain other industrial chemicals, dusts, or fibers
- smoking
- Staphylococcus aureus, a common bacterium, in the nasal passages
People with occupational exposure to silica and other industrial chemicals, such as those who work in agriculture or construction, may be at a particularly increased risk of AAV.
Certain external factors may acutely trigger AAV symptoms in a person with existing risk factors. Potential environmental triggers of AAV symptoms include:
- recent vaccination
The link between infections and AAV has long been suspected. S. aureus is the most well-studied disease-causing microbe that may contribute to AAV and is most closely linked to GPA.
Vaccines have only rarely been connected to AAV, and are still recommended for most people.
Geographical location and the current season may also influence the onset of AAV, though the evidence is inconclusive.
Drug-induced AAV
Certain medications can also cause AAV. It’s thought that these drugs may affect the immune system in ways that promote the development of ANCAs, but the mechanism is not fully established.
Drug-induced AAV and other types of AAV have similar symptoms, but other elements differ.
| Drug-induced AAV | Other AAV types |
|---|---|
| May be related to one or more ANCA types | Typically related to only one ANCA type |
| More common in females | Roughly equal in males and females |
| More common in younger patients | More common in older patients |
| Typically has a milder clinical course and a better prognosis | Typically has a severe clinical course and a worse prognosis |
| Long-term maintenance therapy may not be necessary after discontinuing the medication linked to AAV | Long-term maintenance therapy is often necessary |
A wide range of substances have been potentially linked to AAV. Some of the most common ones include:
- propylthiouracil and other medications used to treat an overactive thyroid
- hydralazine, which is used to treat high blood pressure and heart failure
- cocaine, particularly when contaminated with the veterinary drug levamisole
- certain antibiotics
- certain biologic immunosuppressive medications
Who is more likely to develop AAV?
AAV is primarily a disease of older adults and is most often diagnosed in people ages 55-75. Of the three AAV types, MPA tends to be diagnosed at older ages (65-75 years), while EGPA is the most likely to be diagnosed in younger adults. Childhood-onset AAV is rare.
Similar overall AAV rates are found in male and female patients, although some studies show a slight male predominance.
AAV is most frequently reported among Caucasians, although it occurs across ethnicities.
What is not known about AAV
As with many autoimmune diseases, AAV is unpredictable and lacks a single clear cause. It is not easy to predict who will develop the disease. Researchers are still working to better understand what causes autoimmune vasculitis. Some areas of ongoing research include:
- other potential causes and triggers of AAV
- why AAV sometimes develops without ANCAs
- why people with risk factors don’t always develop AAV
- if AAV can be prevented
In the future, this research may help resolve uncertainty about why AAV occurs and offer new avenues for developing AAV therapies.
ANCA Vasculitis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by 

