Rituxan Shows Promise in GPA Patients with Severe Eye Inflammation Symptoms
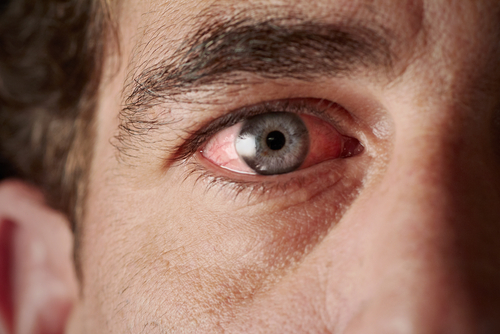
Patients with granulomatosis with polyangiitis (GPA) — formerly known as Wegener’s granulomatosis — with severe inflammation in the eyes may significantly benefit from the anti-CD20 antibody Rituxan (rituximab).
In the study, “Rituximab Induction and Maintenance Treatment in Patients with Scleritis and Granulomatosis with Polyangiitis (Wegener’s),” published in Ocular Immunology and Inflammation, Rituxan effectively induced remission of eye inflammation and prevented relapses in GPA patients.
Severe inflammation of the white external coating of the eye (sclera), known by the medical term scleritis, is commonly associated with other illnesses like GPA or rheumatoid arthritis. Indeed, GPA-associated scleritis accounts for 3.6 to 8.1 percent of all scleritis cases.
In severe cases of scleritis, aggressive treatment regimens with corticosteroids or immune inhibitors (immunosuppressants) may be required to prevent vision loss or blindness due to sclera’s cell death.
However, such therapies may induce unwanted and potentially harmful adverse side effects. In addition, prolonged use is often associated with resistance, promoting GPA-associated scleritis relapse.
The development of biological agents, such as antibodies with immunosuppressant activities, has provided new tools that can be used to treat GPA patients with fewer side effects and improved efficacy.
Rituxan, currently co-marketed by Biogen and Genentech, is an antibody designed to target a surface protein found in immune B-cells. Its activity depletes B-cells for up to six to 12 months, alleviating symptoms of several autoimmune diseases, including rheumatoid arthritis (RA), GPA, and other ANCA-associated vasculitis.
The U.S. Food and Drug Administration approved the use of Rituxan initially for the treatment of relapsed or refractory non-Hodgkin’s lymphomas, and later extended its approval for chronic lymphocytic leukemia, RA, GPA, and microscopic polyangiitis (MPA).
Now, researchers from the Massachusetts Eye Research and Surgery Institution (MERSI) evaluated the safety and efficacy of Rituxan for long-term management of GPA-associated scleritis.
The study enrolled nine GPA patients whose scleritis did not respond to standard immunosuppressive treatment or who did tolerate it. They received two 1,000 mg doses of Rituxan, or four 375 mg doses, as an induction treatment, followed by maintenance Rituxan. In total, 12 eyes were treated.
All treated eyes showed complete remission upon Rituxan therapy, with no symptoms of scleritis. The patients showed clear clinical improvements a median of two months after the first treatment session. Eight patients presented a stable response even after six months.
These positive effects allowed patients to reduce dosage or even discontinue corticosteroid treatment.
No major adverse events were reported during the trial, with only five eyes in four patients showing a mild reduction in visual ability.
“As most GPA relapses occurred after the first two years, [Rituxan] maintenance therapy might be extended over a period of more than 18 months, to decrease the late relapse rate. However, the best regimen for [Rituxan] maintenance treatment for GPA and GPA-associated scleritis has not yet been defined,” the researchers wrote.
“[Rituxan] is a promising option in the management of GPA associated with refractory scleritis and maintenance B-cell depletion might favor long-term remission,” they added.